Enzyme inhibition is the hindrance of the enzyme activity by an enzyme inhibitor. An inhibitor is a substance, other than the enzyme, that attaches itself onto the enzyme thereby inhibiting the enzyme affinity to the substrate. The inhibitor attaches itself to the binding site of the enzyme rendering the enzyme incompatible to the substrate since the enzymes are specific to the substrate onto which they bind to catalyze metabolic processes.
Enzyme inhibition can lead to allergies when exposed to certain types of foods. The inability to produce the right enzyme for substrate metabolism may lead to complex problems such as lactose intolerance.
There are three types of enzyme inhibition namely:
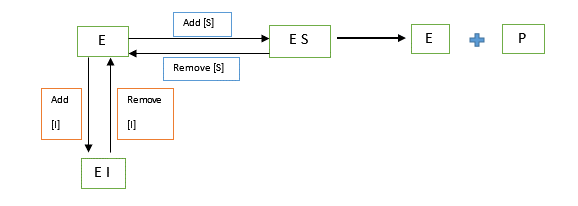
In this type of inhibition, the inhibitor binds itself to the catalytic site and competes with the substrate for the same binding site. The inhibitor has structural similarity to the substrate. It reduces enzymes by forming Enzyme-Inhibitor complex and consequently;
Vmax remains unchanged since at high levels of substrate concentration [S], all the inhibitor is displaced by the enzyme.
Km is increased since higher substrate concentrations are required to reach maximum velocity.
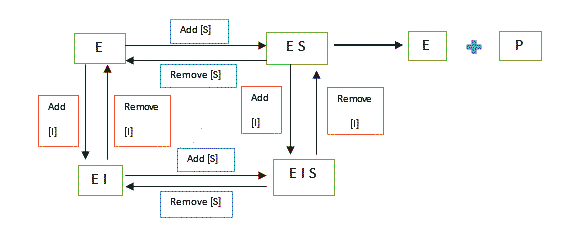
The inhibitor binds to both the Enzyme [E] and the Enzyme-Substrate [ES] complex. The inhibitor need not to be structurally similar to the substrate. As a result of the action of this type of inhibitor, we make these two observations:
Vmax reduces since, at high substrate concentration levels, the inhibitor is still bound.
Km increases since higher substrate concentration [S] is necessary to reach the lower maximum velocity.
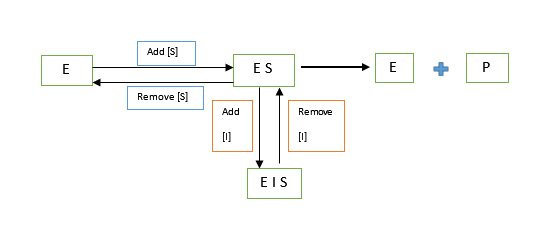
Here, the inhibitor binds directly to the Enzyme-Substrate [ES] complex and not necessarily at the active binding site. The attachment distorts the physical conformation of the enzyme making it unfit for its substrate hence reducing the enzyme’s affinity to the substrate. The inhibitor does not necessarily have to structurally resemble the substrate.
In this type of inhibition, the Vmax is reduced since the amount of ESI formed depends on the concentration of the inhibitor.
Enzyme application in food processing has become industry standard. Endogenous enzymes are critical in food processing. Let us consider an example of each category of enzymes to discuss the enzyme application in fruits and vegetables processing.
Endogenous enzymes naturally exist in food and may affect the quality of the foods (either positively or negatively) by their actions. One of the most cognitive endoenzyme is pectin methyl esterase (PME), which is found in fruits and vegetables. It affects the quality of the final products (such as fruit juices) made from these fruits and vegetables.
This enzyme is applied in texture modification of the thermo-processed fruits and vegetables. This is done by the manipulation of the enzyme using the temperature. For instance, under cold break tomato juice processing, PME rapidly hydrolyses the pectin esters to form methanol, which lowers the pH of the product.
Cold break processing involves heating the tomato to 60°C to activate the PME. The activated enzyme depolymerizes pectin leading to loss of viscosity and cloudiness in the product. This facilitates separation of solid from liquid components of the juice making the process suitable for manufacturing juices such as tomato juice.
Hot break processing on the other hand involves heat inactivation of the PME by heating the juice to 90°C. This results in preservation of the pectin, which promotes and stabilizes cloud and viscosity of the product. Hot break processing is utilized in manufacturing of pastes and gels, e.g. tomato paste.
Exogenous enzymes (exo-enzymes) are those enzymes that you add into the food to promote a desirable effect. An example of the exogenous enzyme is pectin esterase; a pectinase that cracks specific bonds in molecules of pectin.
Pectinase (pectin esterase) is very useful in the manufacturing of fruits and vegetables; it breaks pectin. This results into modification of the texture of fruit juices. For this reason, you can use this enzyme in thinning of pastes/juice concentration and to clarify wines.
You may also add pectin esterase during processing to not only increase the yield of fruit juice concentrates but also to gel the products. Gelation helps in texture modification and improvement of rheological properties of these products.
The key determinants of food quality are established to achieve the high-quality standards that customers expect. Consequently, there is need to implement measures that will ensure that there exists an environment for continuous improvement.
Every consumer expects that the food products s/he uses will not bring any harm to her/his health and that no economic injury will arise from the use of such products. Food safety means that the food is free from any harmful physical, chemical, or any microbial contamination at the point of consumption. Food safety is used in its operational sense e.g. in the canning industry where commercial sterility as applied in low acidity foods is taken to mean the absence of Clostridium botulinum.
To achieve this, there is need to translate the conditions for commercial sterility into a set of heating conditions for a given product in a given package. Optimization of other quality attributes is only achieved after having first considered the basic aspect of safety.
Involves features such as the color, the texture, the taste, the smell, etc. of the food sample under consideration. These are the immediate quality features that an individual recognizes when evaluating a food sample because they are easily perceptible.
An individual is more likely to make a decision on whether to use the food item or not depending on the physically perceptible quality features. There could be undesirable changes in the texture such as the loss of solubility, loss of water holding capacity, hardening or softening.
The food product may also develop a rancid flavor or any other undesirable off-flavors that may not be very appealing to the consumer. Changes in color such as darkening, browning or bleaching may not be very desirable in some products that are associated with a specific color. Sensory appeal thus forms the basis for evaluation and preference by consumers.
Considers the types and contents of nutrients contained in any given food item. It involves a methodical investigation and may require advanced equipment to perform profiled tests to determine the contents of the nutrients. This method also includes investigation of the microbial load in the food item as well as the availability of the nutrients profiled and their digestibility.
Some foods may have certain nutrients in abundance but such nutrients remain largely unavailable for uptake by the human digestive system due to degradation. Storage and processing conditions may lead to loss of certain vitamins, proteins and minerals.
Facilitates handling of the food and makes the food items more functional to the manufacturer and the users.
Some of the features that enhance convenience of food items include:
Enzymatic browning occurs due to the action of polyphenoloxidase enzyme, which brings about the brown coloration in fruits, seafood, and vegetables. The effect of enzymatic browning may lead to reduction in the quality of foods, especially during the post-harvest storage time.
Polyphenols are very unstable; therefore, they react with other components in the food to bring about the brown color effect in foods. The reactions are mostly initiated by injuries to the surface of the food sample.
|
Source |
Phenolic substrates |
|
Apple |
chlorogenic acid (flesh), catechol, catechin (peel), caffeic acid, 3,4-dihydroxyphenylalanine (DOPA), 3,4-dihydroxy benzoic acid, p-cresol, 4-methyl catechol, leucocyanidin, p-coumaric acid, flavonol glycosides |
|
Apricot |
isochlorogenic acid, caffeic acid, 4-methyl catechol, chlorogenic acid, catechin, epicatechin, pyrogallol, catechol, flavonols, p-coumaric acid derivatives |
|
Avocado |
4-methyl catechol, dopamine, pyrogallol, catechol, chlorogenic acid, caffeic acid, DOPA |
|
Banana |
3,4-dihydroxyphenylethylamine (Dopamine), leucodelphinidin, leucocyanidin |
|
Cacao |
catechins, leucoanthocyanidins, anthocyanins, complex tannins |
|
Coffee beans |
chlorogenic acid, caffeic acid |
|
Eggplant |
chlorogenic acid, caffeic acid, coumaric acid, cinnamic acid derivatives |
|
Grape |
catechin, chlorogenic acid, catechol, caffeic acid, DOPA, tannins, flavonols, protocatechuic acid, resorcinol, hydroquinone, phenol |
|
Lettuce |
tyrosine, caffeic acid, chlorogenic acid derivatives |
|
Lobster |
tyrosine |
|
Mango |
dopamine-HCl, 4-methyl catechol, caffeic acid, catechol, catechin, chlorogenic acid, tyrosine, DOPA, p-cresol |
|
Mushroom |
tyrosine, catechol, DOPA, dopamine, adrenaline, noradrenaline |
|
Peach |
chlorogenic acid, pyrogallol, 4-methyl catechol, catechol, caffeic acid, gallic acid, catechin, dopamine |
|
Pear |
chlorogenic acid, catechol, catechin, caffeic acid, DOPA, 3,4-dihydroxy benzoic acid, p-cresol |
|
Plum |
chlorogenic acid, catechin, caffeic acid, catechol, DOPA |
|
Potato |
chlorogenic acid, caffeic acid, catechol, DOPA, p-cresol, p-hydroxyphenyl propionic acid, p-hydroxyphenyl pyruvic acid, m-cresol |
|
Shrimp |
tyrosine |
|
Sweet potato |
chlorogenic acid, caffeic acid, caffeylamide |
|
Tea |
flavanols, catechins, tannins, cinnamic acid derivatives |
Common polyphenols that cause browning (source, www.food-info.net/uk/colour/enzymaticbrowning.htm)
Different enzymes have different inactivation temperatures.
|
Enzyme |
Effect caused |
Inactivation temp. (°C) |
|
Lipolityc acyl hydrolase |
rancidity |
~ 75 |
|
Lipoxygenase |
rancidity |
~ 80 |
|
Polyphenoloxidase |
browning |
~100 |
|
Peroxidase |
deterioration |
~135 |
Approximated inactivation temperatures of some enzymes
iii) Acidulants such as citric acid and phosphoric acid act by lowering medium pH.
A form of browning effect in foods that is initiated by either Caramelization or Millard reaction. Caramelization happens when the sugars in foods are exposed to high temperatures. Water is removed from the sugar whose isomers are them polymerized. Millard reaction occurs when the amino acids react with reducing sugars in the presence of heat (can also happen in the absence of heat).
Here, the carbonyl group of the sugar reacts with the amino group in the amino acid to produce melanoidins responsible for various flavors and colors. Different amino acids produce different flavor components, which makes Millard reaction a key component in the flavoring industry.
Ascorbic acid and metapolyphenols can also contribute to non-enzymatic browning. Heating leads to formation of furfurals (isomers), which then polymerize to produce the brown color observed as the browning effect. Millard reactions may reduce the nutritive value of foods. For instance, toasting bread may lead to loss of up to half the protein efficiency ratio of bread.
Lipid hydrolysis is the breakdown of lipids by addition of water to produce glycerol and fatty acids. This usually happens in an enzyme (lipolytic enzymes such as lipases) mediated process. The free fatty acids produced by the hydrolysis process may lead to rancidity, which is not desirable in most cases, as the resultant food product will have a poor sensory appeal.
If the hydrolysis occurs in an aqueous sodium hydroxide medium, soap is formed. Foods most susceptible to hydrolysis include edible oils, margarine, butter, fried foods (potato chips), roasted nuts, dried soups, broths, seasonings, milk, dried meat, frozen fish, etc.
Store foods in a cool and dry place to avoid non-enzymatic browning. Eliminate all lipases to avoid the enzymatic action on the food product.
The presence of oxygen in lipid foods will initiate an oxidation process. When the oxidation process is initiated, it will lead to deterioration of the fat phase of the food. Deterioration of fatty foods is a phenomenon commonly referred to as rancidity.
Uncontrolled oxidation of lipids could lead to formation of several cytotoxic and mutagenic compounds that are harmful to the body. However, not all oxidative reactions in lipids are bad since the β-oxidation process utilizes lipids to produce energy. Eicosanoids, which are signaling substances in the body, are also produced through lipid oxidation.
Involves the alteration of the normal protein structure and a possible destruction of the secondary conformations of such structures. Some protein denaturation processes are irreversible e.g. cooking an egg. Heat, alcohol, heavy metal salts, acids and bases are some of the agents of protein denaturation.
Protein cross-linking occurs when the reactive ends of two or more functional groups (such as amines and sulfhydrils) on proteins covalently join to form two or more biomolecules.
The resultant biomolecules may exhibit increased/reduced solubility, stability, aggregation, etc and may cause food poisoning when consumed.
Protein hydrolysis involves breakdown of proteins into its constituent amino acids through enzymatic and chemical methods. The most common chemical for protein hydrolysis hydrochloric acid while most enzymes used for protein hydrolysis are derived from animal (e.g. pepsin) and plant (e.g. ficin from fig tree) sources.
After hydrolysis, the resultant peptides and amino acids may form poisonous substances rendering the food unsafe for consumption.
Pigments contribute to the colour of the food and as a result, the perceived quality of the food. Pigments in food can be naturally occurring such as carotenoids or added in the form of synthetic organic dyes. Natural pigments are unstable and take part in different reactions. Pigments such as carotenoids contain a high number of conjugated double bonds that are reactive electron-rich systems susceptible to reaction with electrophilic compounds.
Most carotenoids are singlet oxygen quenchers. Singlet oxygen interacts with the carotenoid to give triplet states of both the energy of an excited carotenoid is dissipated through vibrational interaction with the solvent to recover the ground state molecules. Carotenoids are the most occurring quenchers of singlet oxygen. They may participate in the propagation step of the oxidation process as chain breaking antioxidants that scavange reactive peroxyl radicals (van den Berg et al., 2000).
However, carotenoids serve this function best at low oxygen tensions. At higher oxygen levels, a carotenoid intermediate radical might add oxygen to form carotenoid peroxyl radicals such as Car-OO, which could act as prooxidants initiating the process of lipid peroxidation (Massoud, Nikbakht and Massoud, 2017). On the other side, radicals and peroxides, occurring in food as a result of lipid oxidation, accelerate oxidative degradation of carotenoid pigments, which leads to formation of epoxides. Lipoxygenase involved in the decay of vegetable matter may also cause the destruction of carotenoids.
Chlorophyll pigments are also unstable and may be used as an indicator of health and ripeness of different plant material as well as of processing condition. The degradation of chlorophyll may occur within a few or several weeks and is dependent on the pH and temperature. Chlorophyll transforms by the loss of Magnessium (Mg) in dilute acids or replacement of Mg by other divalent metals. The removal of Mg gives olive-brown pheophytin a and b. Replacing Mg by Iron (Fe) yields grayish-brown compounds while Copper(Cu) or Zinc (Zn) retains the green colour.
These changes in colour are strongly associated with the quality and freshness of fruits and vegetables (Tomasik, 2007).
Anthocyanins are water soluble pigments in plants that are composed of aglycone (anthocyanidin) esterified to one or more of five sugars (glucose, rhamnose, xylose, galactose and arabinose). Decolourization of the pigments is dependent on pH. Anthocyanins are stable on low pH characterised by a red-purple colour. However, under basic pH they are light blue in colour.
Polysaccharides are important complexing agents for inorganic and organic gases, liquids, and solids. Retrogradation is a very common reaction of gels of starch polysaccharides. It leads to enhanced molecular-weight systems. Amylose gels retrograde within hours, whereas retrogradation of amylopectin takes days and even weeks. This process is manifested by dendrite formation in the gel and in bread by bread staling and water expulsion.
This phenomenon is due to the orientation of chains of polysaccharides with respect to one another to aggregate with involvement of intermolecular hydrogen bonds. The retrogradation affinity depends on the starch variety and decreases in the following order: potato > corn > wheat > waxy corn starch. Evidently, the retrogradation rate and nature of the formed amylose crystals depend on the starch source, amylose-to-amylopectin ratio, and storage temperature. Low temperatures around the freezing point and polar gel additives favour retrogradation. Retrograded starch is utilized as a component of low-calorie foods.
Polysaccharides undergo hydrolysis; reduction to alcohols, oxidation to aldehydes, ketones, and carboxylic acids; esterification with inorganic (sulfuric, phosphoric, nitric, boric, and sillylic) and organic acids; etherification; acetylation with aldehydes; halogenation with the same reagents as mono- and disaccharides; ammination (usually via halogenated polysaccharides); carbamoylation with acrylamides or isocyanates; and metallation.
Enzymes are used in the form of pure or semi-pure preparations or together with their producers, that is, microorganisms. Currently, semisynthetic enzymes are also in use. Alcoholic fermentation is the most common method of utilization of monosaccharides, sucrose, and some polysaccharides, such as starch.
Hydrolysis of polysaccharides with alpha-amylase, so-called alpha-amylolysis, is the common way of hydrolysis of starch to maltodextrins. Recently, it has been shown that such reaction can be stimulated by illumination of the enzyme with linearly polarized light. The amylolysis can be accelerated by several orders (Fiedorowicz & Chaczatrian, 2003).
Lactic acid fermentation is another important enzymatic process. Lactic acid bacteria metabolize mono- and disaccharides into lactic acid. This acid has a chiral center; thus either D(–), L(+), or racemic products can be formed. Among lactic acid bacteria, only Streptococcus shows specificity in the formation of particular enantiomers, and only the L(+) enantiomer is produced.
Enzymatic reduction of glucose-6-phosphate into inositol-1-phosphate with cyclase and reduced NAD coenzyme, followed by hydrolysis with phosphatase, presents another nondegrading enzymatic process proceeding on hexoses. Inositol resulting in this manner from its phosphate, plays a role in the growth factor of microbes. Its hexaphosphate, phytin, resides in the aleurone layer of wheat grains.
There are also known bacteria that polymerize mono- and oligosaccharides.
Leuconostoc mesenteroides polymerizes sucrose into dextrin—an almost linear polymer of 400 or more α-D-glucose units. Dextran is also generated in frozen sugar beets. This causes difficulties in sugar manufacturing if the beets have to be stored at low temperature.
Dextran serves as a blood substitute and chromatographic gel (Sephadex). Other polysaccharides synthesized by bacteria are levan, a polymer of β-D-fructose, pullulan, a polymer of α-D-glucose, and xanthan gum, a polymer of β-D-glucose and α- and β-D-mannoses.
Starches can be modified to suit processing in food technology. The functional group reactivity in polysaccharides is hindered to a great extent by their large structure. The potential reaction sites are unavailable for many reactions as they reside on the inside of the macrostructure. These sites may also be involved in the formation of intra- and intermolecular hydrogen bonds, vital for macrostructure properties.
Many reactions of polysaccharides are governed by heterogeneity of the reaction system. Only random reacting polysaccharides are solvent soluble. Among polysaccharides utilized either on or after transformation, hemicelluloses are exclusive. They are water soluble or they swell.
Problems of solubility and compactness are encountered, particularly in cellulose and starch, which are fibrillar and granular, respectively. The reactivity in terms of the rate and degree of transformation can be controlled by either application of suitable reagents or loosening of the compact structure with involvement of a physical action.
Pasting and gelatinization of starch by heating it in water or immersing it in aqueous alkali delivers pregelatinized starch. Because such processing breaks several intra- and intermolecular hydrogen bonds, pregelatinized starch is easily water soluble and chemically more active. When granular starch is passed under pressure through narrow nozzles, so-called α-starch (nanostarch) with improved solubility in water is formed.
Fresh fruits and vegetables proceed to ripen and eventually decay through a process of senescence; normal aging of the produce. They continue to be a living entity, and continue to respire (analogous to breathing) in that oxygen is consumed and carbon dioxide and water vapour are generated. This aging process continues until the product dies and starts to decay.
Bananas, for example, are typically harvested green, ripen to a yellow fruit, and finally over ripen first to a speckled brown, then to primarily brown, and ultimately a black appearance. This ripening process is coded into the genetic make-up of the produce. This can be partially controlled by temperature, composition of the environment through controlled or modified atmosphere, and by scrubbing triggering agents such as ethylene which initiates the ripening of bananas and other fruits (ITC, 2002).
Nitrosation depends on the pH with the optimum pH values being between 2 and 4. The nitrous acid generated in low acid foods from endogenous or added nitrates decompose easily too yield the nitrosating agents nitrous anhydrate and the nitrosonium ion.
The reaction of these compounds with the secondary and tertiary amines contained in many foods lead to carcinogenic N-nitrosamines.
The secondary and tertiary amines are responsible for the fishy odours from food making is undesirable. Microorganisms such as Pseudomonas, Bacillus and Clostridium can take part in these reactions. The decrease in pH in food results in increase in decarboxylase activity of bacteria. The bacteria produce more decarboxylases as part of their protective mechanism (Stadnik & Dolatowski, 2010).


Comments