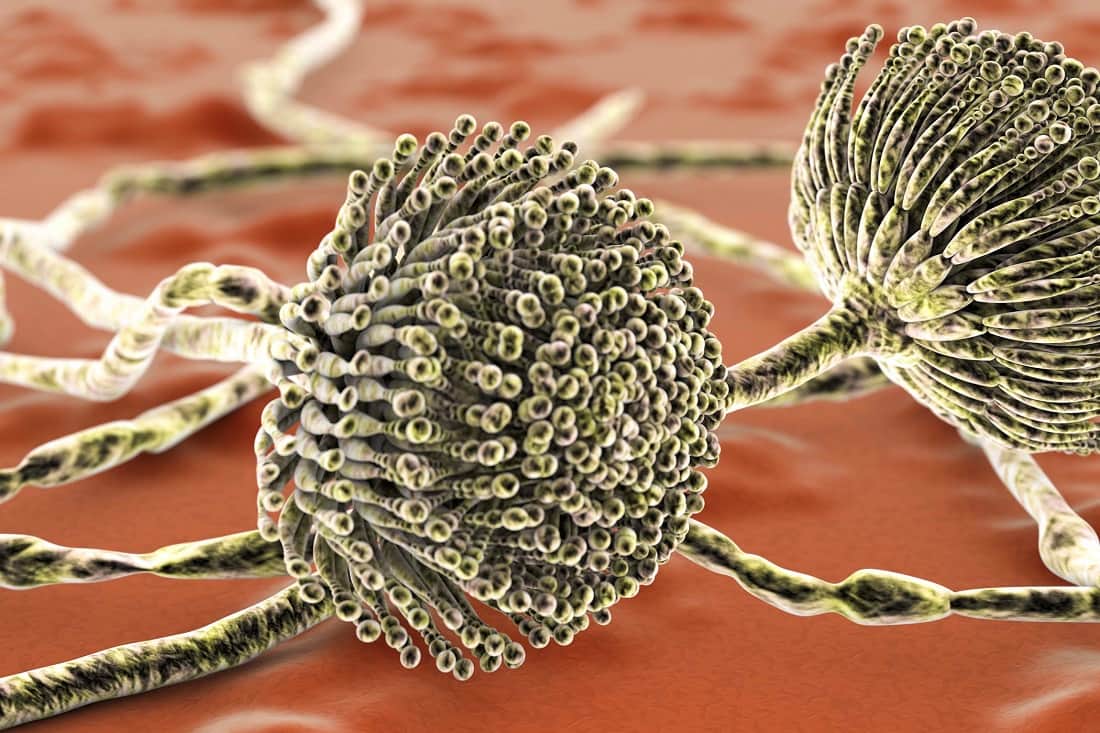
Mycotoxins are poisonous fungal metabolites of which, aflatoxins are the most prominent. They are produced when someone ingests fungal-infested food and can be very toxic, even lethal. Aflatoxins are the biggest threat of all forms of fungal intoxication on food.
Several fungi that produce these mycotoxins and they target many foods such as cereals, fodder crops, and even hay.
The following factors favour their growth, multiplication, and reproduction:
These conditions create a perfect environment for the thrift of these fungi, which spoil food by rendering the food not fit for consumption. They are produced in cereals in the field and during storage when the temperatures are high and the levels of humidity are elevated.
This is the disease that arise as a result of aflatoxin poisoning. It can occur in both man and animals and it produces chronic signs in acute doses. It was first discovered in early 1960 in turkeys and ducklings.
It is caused by the fungus Aspergillus flavus. The common toxins produced include B1, B2, G1, and G2, which are direct action toxins. Contamination of commercial cereals can be catastrophic.
Indirect toxins such as M1 and M2 can also affect man by consuming products (e.g. milk) from animals with afflatoxicosis. Due to their mode of action, they are generally labelled as factors.
Aflatoxin is thermo stable but alkaline labile.
The most vulnerable species are all animals with pets taking the heaviest blow due to their small sized bodies. They catch the poison from contaminated food.
In larger species, the young ones will be adversely affected. All humans suffer the same severity unless one has compromised immunity.
However, aflatoxins in meat is generally not considered a very big problem.
Since aflatoxin poison affects the liver, other symptoms arise from the liver failure. Aflatoxins are carcinogenic and induce tumour formation (in chronic cases).
The observable symptoms include:
Clinical signs and health history can be associated with mouldy food/feed. To confirm the presence of the toxin, it is isolated from the feed and animal tissues (especially the liver).
Proper harvesting techniques and rapid post-harvesting drying of cereals.
Control of temperature and humidity during storage and transportation of cereal foods. Use fungicides.
Monitor the hygiene of animal feed. Control mould growth on the animal feed (e.g. hay and silage). Do not feed animals on aflatoxin-infested cereals.
Animals get poisoned via two major routes:
By ingesting the preferred poison
Through secondary poisoning by ingesting the poisoning agents. The plants they feed on may also be toxic, causing either primary or secondary poisoning.
Only a qualified person should do the diagnosis since the outcomes do have legal implications.
Records are important since history plays a critical role in diagnosis, especially in cases where the feed is the suspect.
Clinical signs are also important since most poisoning cases usually end in fatality. Post mortem is advised.
Laboratory analysis will confirm the suspicions.
Biological poisons tend to be plants, usually the drought resistant types.
Chemical poisons may include:
Physical poisoning agents include:
Food animals (animals generally consumed as food) may cause secondary poisoning. It will pass the poison to the secondary consumer through their tissue.
For that reason, here is what you need to do when checking for an animal to eat:


Comments